Our Clinical Vision: AVIM Therapy for Hypertensive Heart Disease
Orchestra BioMed recently announced multiple scientific presentations highlighting the robust body of existing clinical findings from the Atrioventricular Interval Modulation (“AVIM”) Therapy program at the Technology Heart Failure and Therapeutics (“THT”) and Cardiovascular Research Technologies (“CRT”) 2026 Meetings. These discussions reinforce growing clinical interest in our novel, device-based approach to treating hypertension and reducing associated cardiovascular risk.
Hypertension is not just about elevated blood pressure; it’s a progressive cardiovascular condition that can drive structural and functional changes in the heart. As hypertension persists and progresses, it can transition to hypertensive heart disease and have broader impact on cardiovascular health and the function of critical organs such as the kidneys. Hypertensive heart disease can lead to higher risk of major events like heart attack and stroke as well as play a critical role in the development and progression of heart failure, particularly heart failure with preserved ejection fraction, also known as HFpEF. The scientific presentations at THT and CRT help frame the potential role of AVIM Therapy to address unmet treatment needs of patients with hypertensive heart disease and HFpEF.
David E. Kandzari, MD, Chief of the Piedmont Heart Institute and Co‑Principal Investigator of the BACKBEAT global pivotal study, expressed excitement for the transformative potential of AVIM Therapy to improve clinical outcomes for these patients:
“AVIM Therapy has the potential to offer a new treatment paradigm that leverages active implantable devices with a long, established clinical history. Pilot study clinical and mechanistic results demonstrate that modulation of the atrioventricular interval drives hemodynamic and autonomic nervous system function impact that could help patients with HFpEF, a population with limited therapeutic options.”
Dr. Kandzari’s commentary on the potential benefit of AVIM Therapy for patients with heart failure with preserved ejection fraction (HFpEF) is supported by results from the MODERATO I pilot study, which demonstrated significant reductions in heart rate and end-diastolic volume at 3 and 24 months, while maintaining stable end-systolic volume and ejection fraction.
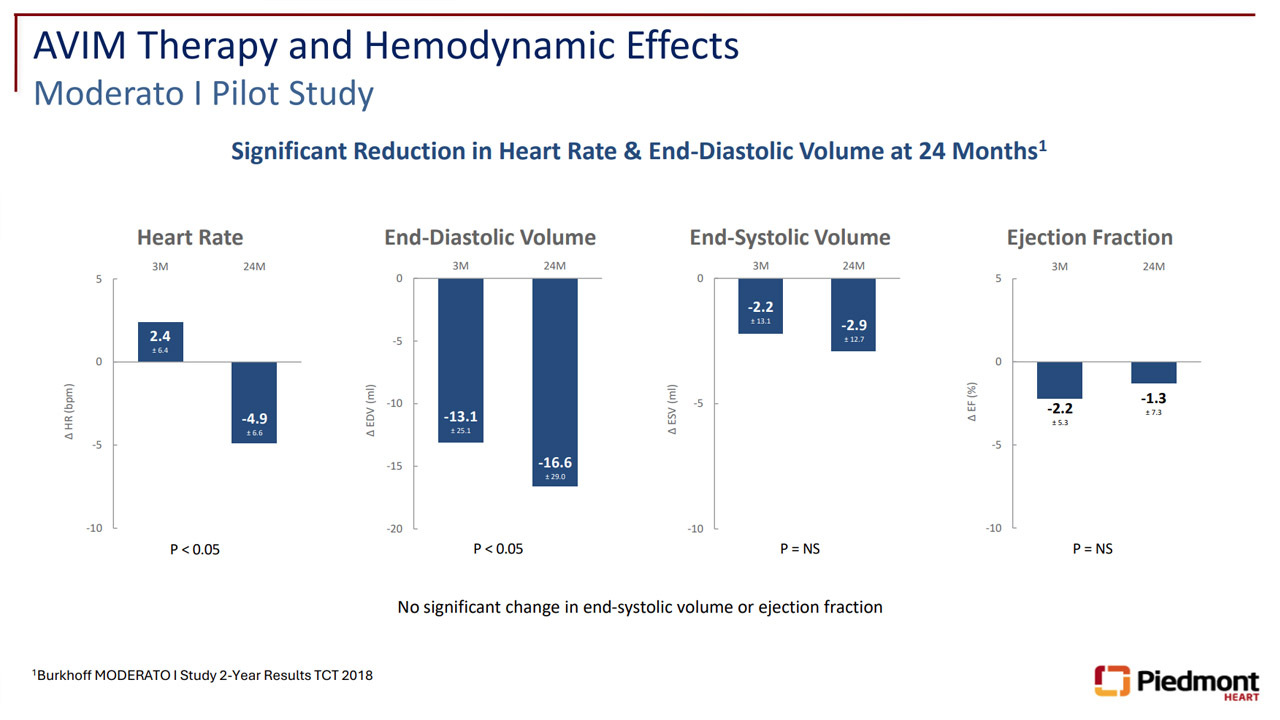
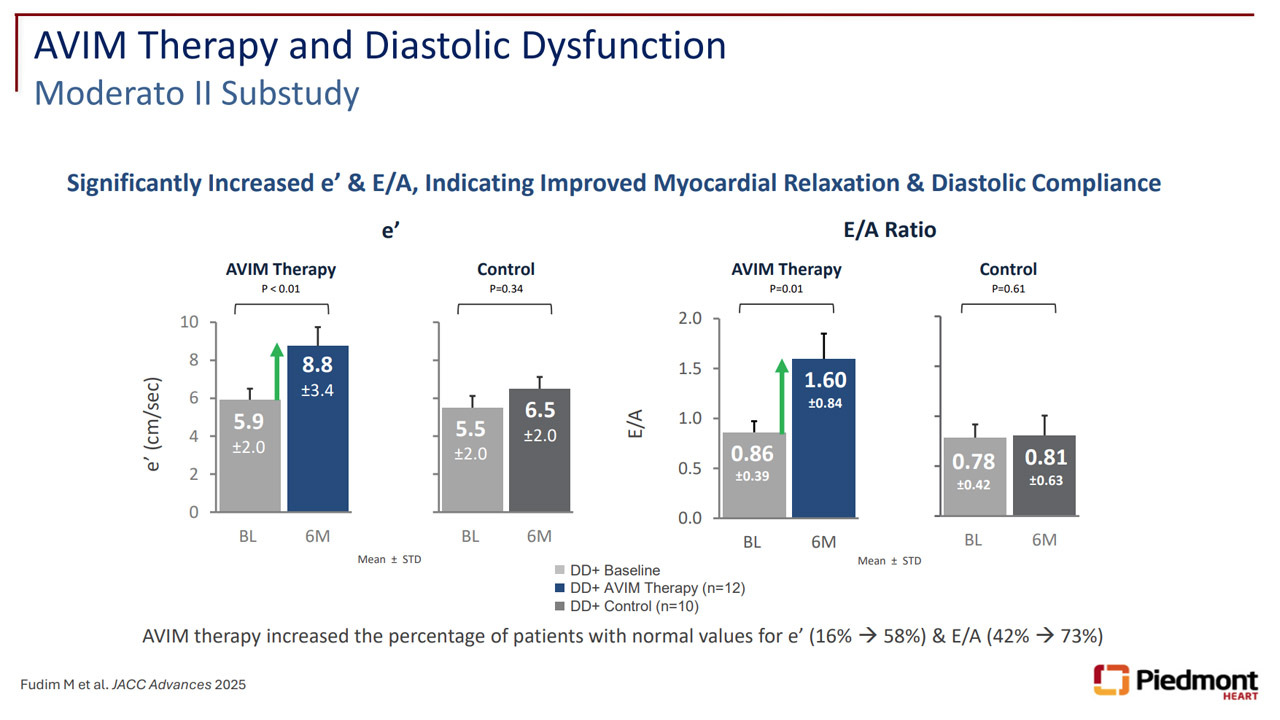
Additional data from a MODERATO II sub-study demonstrated improvements in diastolic function, including increased e′ and E/A ratio, extending the clinical potential of AVIM Therapy beyond blood pressure reduction toward possible relevance in prevention of comorbid conditions such as stroke, MI and heart failure.
See Dr. Kandzari’s full THT presentation here.
A Differentiated Bioelectronic Approach to Hypertension
Orchestra’s initial target population for AVIM Therapy is in patients who have uncontrolled hypertension and are indicated for a dual-chamber pacemaker. Hypertension is highly prevalent in this population which is typically comprised of older patients with increased risk of progression to hypertensive heart disease and other cardiovascular complications. Despite the availability of numerous antihypertensive medications, many patients remain inadequately controlled.
AVIM Therapy leverages programmable cardiac pacing patterns within an implanted pacemaker system to immediately and substantially reduce blood pressure while regulating autonomous nervous system responses. By intentionally modulating atrioventricular interval timing, the therapy is designed to change cardiac filling dynamics to reduce systolic blood pressure through a mechanism that acts directly on cardiovascular physiology rather than through pharmacologic pathways.

Because the therapy is delivered using a standard dual-chamber pacemaker system, it has the potential to integrate seamlessly into routine cardiac device care while introducing a novel hypertension management strategy that does not burden patients with medication adherence and tolerance.
Clinical Findings Supporting the AVIM Therapy Mechanism of Action
Pilot studies evaluating AVIM Therapy have shown clinically meaningful and statistically significant reductions in systolic blood pressure in pacemaker‑indicated patients with hypertension, with effects observed immediately upon activation of the therapy and sustained through long‑term follow‑up. Notably, these therapeutic effects were observed in a population of patients that predominantly suffer from isolated systolic hypertension (“ISH”), a challenging-to-treat and more dangerous form of hypertension that is often underrepresented in clinical research.
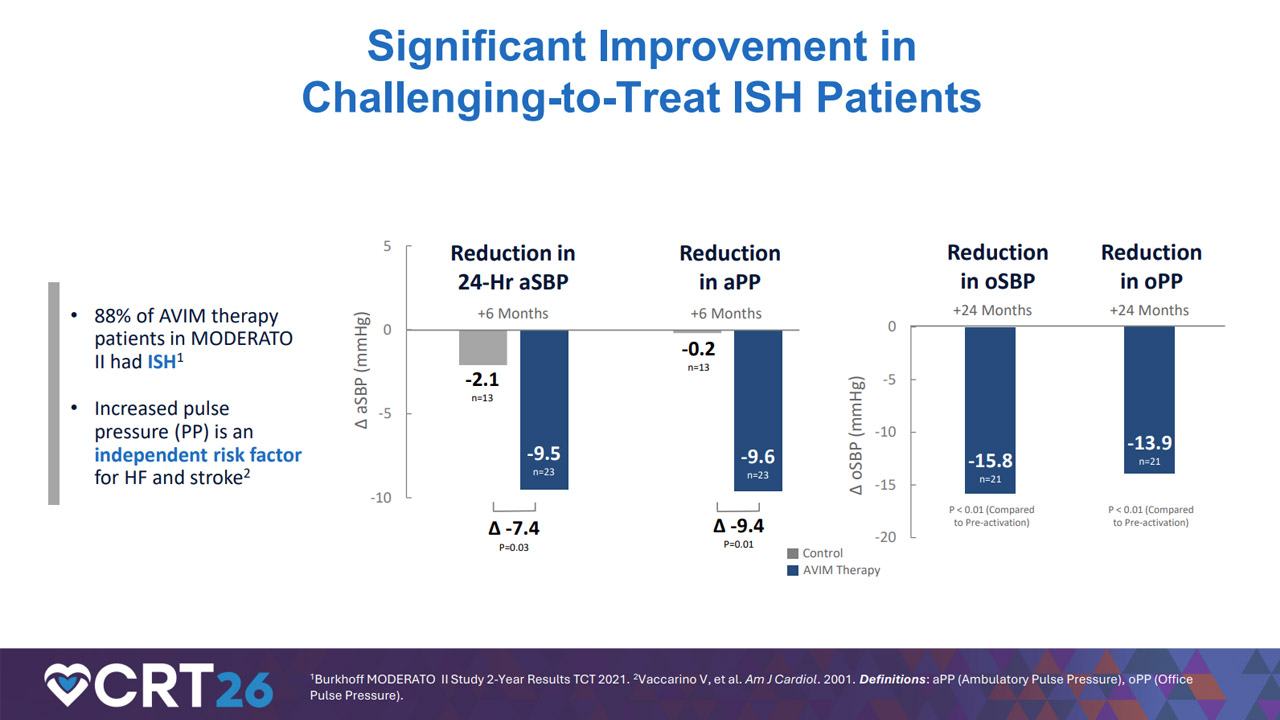
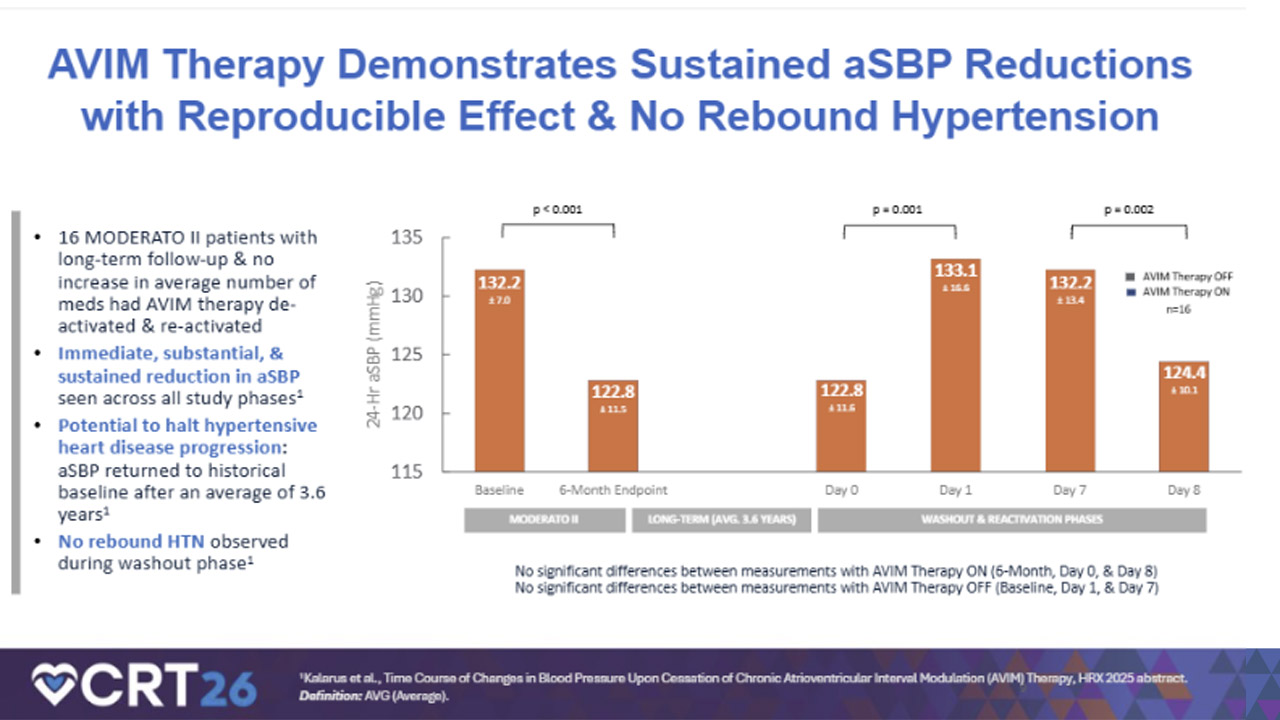
Dan Burkhoff, MD, PhD, Director of Heart Failure, Hemodynamics and Mechanical Circulatory Support Research at the Cardiovascular Research Foundation, and Chairman of the Clinical Steering Committee at Orchestra Biomed, highlighted the durability and reproducibility of effect observed in an analysis of a washout period, or brief time of deactivation, after long-term treatment with AVIM Therapy:
“In pilot studies, AVIM Therapy demonstrated an immediate reduction in systolic blood pressure that starts upon treatment activation and persists through nearly four years of follow‑up. Long‑term washout pilot study data demonstrating reproducible blood pressure reduction without evidence of disease progression further highlight the unique potential clinical utility of this therapy.”
See Dr. Burkhoff’s full CRT presentation here.
Taken together, these data highlight the importance of sustained blood pressure control in older, comorbid patients, without requiring additional medication adherence, for reducing the risk of stroke, myocardial infarction, and heart failure.

Looking Ahead: Focused on Clinical Execution
AVIM Therapy is currently being evaluated in the BACKBEAT global pivotal study (“BACKBEAT Study”), a randomized clinical trial designed to assess the efficacy and safety of the therapy in patients with uncontrolled hypertension who require a dual‑chamber pacemaker. Orchestra is conducting the BACKBEAT Study as part of a strategic collaboration with Medtronic (NYSE: MDT), the global leader in cardiac pacing therapy. Medtronic has exclusive rights to commercialize AVIM Therapy for the treatment of uncontrolled hypertension in patients indicated for a pacemaker and a right of first refusal to expand rights to treatment of patients not indicated for a pacemaker.
The BACKBEAT study is intended to provide the clinical evidence required to support potential regulatory approval and subsequent commercial adoption of AVIM Therapy.
As Orchestra BioMed continues to advance the BACKBEAT Study, the company is also focused on continuing to build and strengthen the scientific foundation supporting AVIM Therapy in broader populations of patients beyond those indicated for a pacemaker. Through continued research, strategic collaboration, and engagement with the global cardiology community, Orchestra BioMed is working to maximize the full potential AVIM Therapy as a differentiated device‑based therapy for hypertensive heart disease.
- Team OBIO
Published March 2026
Forward-Looking Statements
Certain statements included in the content distributed by Orchestra BioMed Holdings, Inc. (the “Company”) to which this disclaimer relates (including, but not limited to, visual, auditory, or written statements accessible through the links included in the Company’s digital communication) (the “Content”) that are not historical facts are forward-looking statements for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995. Forward-looking statements generally are accompanied by words such as “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “intend,” “expect,” “should,” “would,” “plan,” “predict,” “potential,” “seem,” “seek,” “future,” “outlook” and similar expressions that predict or indicate future events or trends or that are not statements of historical matters. These forward-looking statements include, but are not limited to, statements relating to enrollment, timing, implementation and design of the Company’s ongoing pivotal trials, realizing the clinical and commercial value of AVIM Therapy, the potential safety and efficacy of the Company’s product candidates, and the ability of the Company’s partnerships to accelerate clinical development.. These statements are based on various assumptions, whether or not identified in the Content, and on the current expectations of the Company’s management and are not predictions of actual performance. These forward-looking statements are provided for illustrative purposes only and are not intended to serve as and must not be relied on as a guarantee, an assurance, a prediction, or a definitive statement of fact or probability. Actual events and circumstances are difficult or impossible to predict and may differ from assumptions. Many actual events and circumstances are beyond the control of the Company. These forward-looking statements are subject to a number of risks and uncertainties, including changes in domestic and foreign business, market, financial, political, and legal conditions; risks related to regulatory approval of the Company’s commercial product candidates and ongoing regulation of the Company’s product candidates, if approved; the timing of, and the Company’s ability to achieve expected regulatory and business milestones; the impact of competitive products and product candidates; and the risk factors discussed under the heading “Item 1A. Risk Factors” in the Company’s Form 10-K for the year ended December 31, 2025, which was filed with the SEC on March 12, 2026. The Company operates in a very competitive and rapidly changing environment. New risks emerge from time to time. Given these risks and uncertainties, the Company cautions against placing undue reliance on these forward-looking statements, which only speak as of the date of the Content. The Company does not plan and undertakes no obligation to update any of the forward-looking statements made in the Content, except as required by law.

Strengthening AVIM Therapy’s Regulatory Pathway: A Second FDA Breakthrough Device Designation
Strengthening AVIM Therapy’s Regulatory Pathway: A Second FDA Breakthrough Device Designation Orchestra BioMed Investor ...

From Blood Pressure Reduction to Cardiac Function: What New AVIM Therapy Data Signal
From Blood Pressure Reduction to Cardiac Function: What New AVIM Therapy Data Signal Orchestra BioMed Investor Relations...

Our Clinical Vision: AVIM Therapy for Hypertensive Heart Disease
Our Clinical Vision: AVIM Therapy for Hypertensive Heart Disease Orchestra BioMed recently announced multiple scientific...

When Partnership Drives Progress: Reflections on the Vivasure Journey
When Partnership Drives Progress: Reflections on the Vivasure Journey The recent acquisition of Vivasure Medical by Haem...

2025 Reflections from OBIO CEO
2025 Reflections from OBIO CEO Dear Fellow Shareholders, As 2025 comes to a close, I want to take the opportunity to ref...





